Research summary
Optical technologies applied in the field of diagnostic medical imaging have revolutionized how we understand, detect, and treat diseases over the past decades. My research centers on developing novel diagnostic optical imaging techniques and their clinical translations at the interface of imaging physics, biomechanics, and medicine.
I summarized three significant contributions for my past and current research:
I summarized three significant contributions for my past and current research:
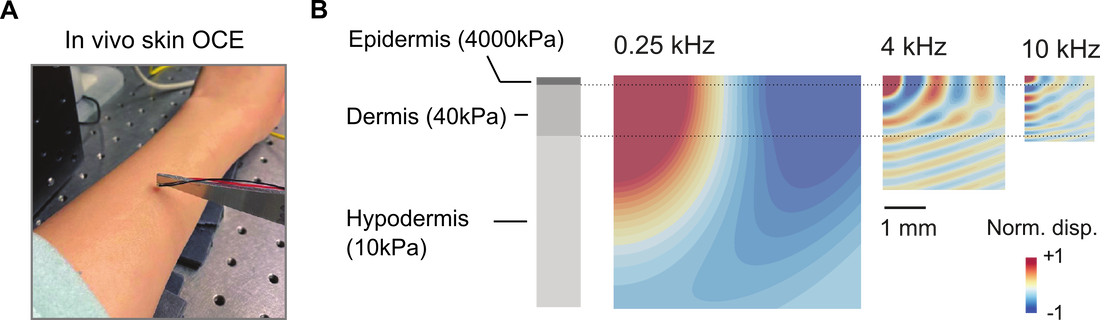
- Ultra-wideband OCE technique from kHz to MHz range. I have developed a novel optical coherence elastography (OCE) technique that extends the upper frequency limit 100x, and improves the spatial resolution 10x (Feng et al., Nature Communications 2023).
- High-resolution stiffness mapping of human skin and cornea in vivo. I have developed a Rayleigh-wave OCE technique and a novel inverse algorithm to characterize the stiffness of in vivo human skin (Feng et al., Acta Biomaterialia 2022), and in vivo human cornea (Feng et al., IEEE BME (pending) and Acta Biomaterialia (pending) with the highest ever lateral and depth resolution.
- Biophysical basis of skin cancer detection using Raman spectroscopy. I have built a Raman biophysical human skin cancer model that classifies skin cancer based on eight Raman active skin constituents (Feng et al., Biomedical Optics Express 2017). My model has been successfully applied to two clinical applications: in vivo skin cancer screening (Feng et al., Journal of Biomedical Optics 2018) and skin cancer surgical margin detection in Mohs micrographic surgery (Feng et al., Biomedical Optics Express 2019; Feng et al., Biophotonics, 2020).
(2019 - Present, Prof. Seok-Hyun (Andy) Yun Lab)
Biomechanical characterization of tissues and clinical translation using optical coherence elastography
Project 1: Ultra-wideband optical coherence elastography (OCE)
|
Conventional shear wave imaging has been limited to the acoustic frequency (~10 kHz). Extending the frequency to the ultrasonic range is challenging due to limited system detection sensitivity.
I developed a ultra-sensitive OCE system that extends the frequency bandwidth from kHz to MHz range. The significance of this work lies in: - Dynamic shear rheological analysis in a previously unexplorable window - High resolution mechanical mapping of intact tissues Featured publication:
|
Fig. 1. (A) Broadband dynamic shear analysis and (B) Depth-resolved stiffness mapping in complex layer-tissues using the ultra-wideband OCE.
|
Project 2: High-resolution stiffness measurement of the human skin and cornea in vivo
Noninvasive measurement of layer-specific structures in tissues and biomaterials has been challenging due to the limited resolution of the measurement tool. To our knowledge, this is the first study that resolves the stiffness of the thin epidermis from the dermis and hypodermis, made possible by using broadband Rayleigh-wave optical coherence elastography.
Resolving layer-specific stiffness of human skin in vivo using broadband Rayleigh-wave OCE.
|
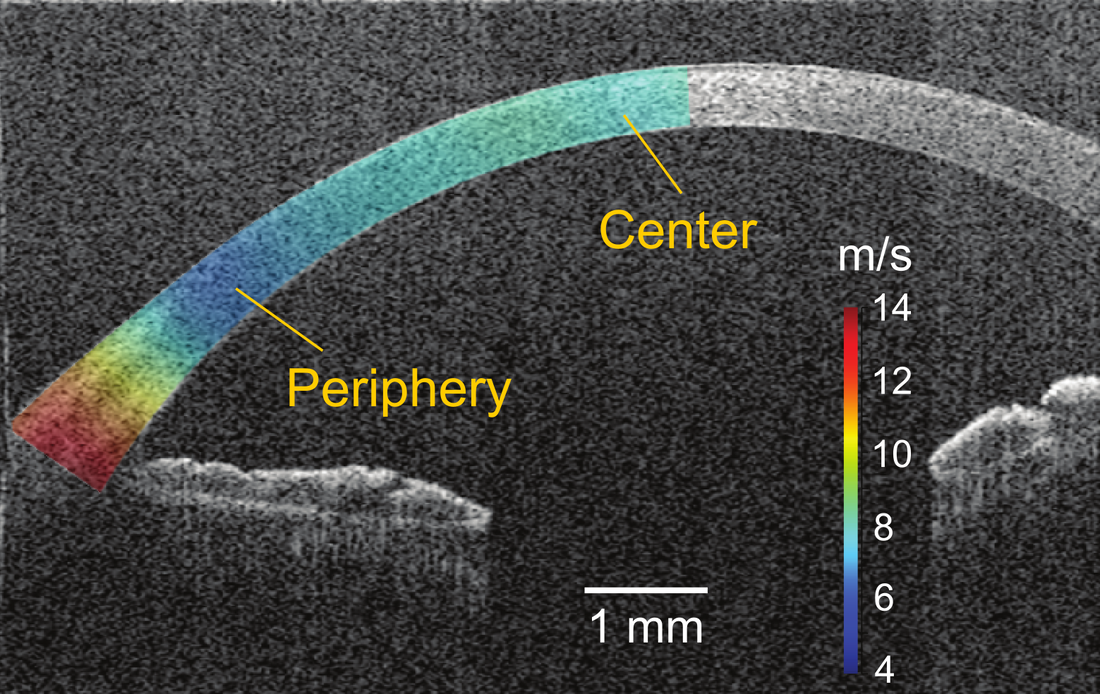
In vivo assessment of corneal biomechanics has been an unmet clinical need in ophthalmology. At present, there are no available experimental or clinical instruments capable of quantitatively assessing corneal elastic properties with the desired spatial resolution. Additionally, existing methods cannot adequately characterize the highly anisotropic, nonlinear, and nonuniform nature of corneal elastic properties. In response to this gap, I developed two in vivo corneal OCE techniques for achieving high-resolution, quantitative measurements of corneal elastic properties. |
High-resolution stiffness mapping in human cornea in vivo.
|
Featured publications:
- Feng X, Li GY, Ramier A, Yun SH. "In vivo stiffness mapping of human cornea using optical coherence elastography", In Bio-Optics: Design and Applications. (pp. JW2B.1). Optica Biophotonics Congress (2023). DOI: 10.1364/BODA.2023.JW2B.1
- Li GY, Feng X (co-first), and Yun SH. "In vivo optical coherence elastography reveals spatial variation and anisotropy of corneal stiffness", arXiv preprint arXiv:2307.04083 (2023). DOI: 10.48550/arXiv.2307.04083 DOI: 10.48550/arXiv.2307.04083
- Li GY, Feng X (co-first), and Yun SH. "Simultaneous measurement of tensile and shear moduli of the human cornea in vivo with S0- and A0-wave optical coherence elastography", arXiv preprient (2023).
- Feng X, Li GY, Ramier A, Eltony AM, and Yun SH. "In vivo stiffness measurement of epidermis, dermis, and hypodermis using broadband Rayleigh-wave optical coherence elastography", Acta Biomaterialia, 146, p.295-305 (2022). DOI: 10.1016/j.actbio.2022.04.030
- Vinas-Pena M, Feng X, Li GY and Yun SH. "In situ measurement of the stiffness increase in the posterior sclera after UV-riboflavin crosslinking by optical coherence elastography", Biomedical Optics Express, 13(10), pp.5434-5446 (2022). DOI: 10.1364/BOE.463600
(2014 - 2019, Prof. James Tunnell Lab)
Noninvasive detection of skin cancer using Raman spectroscopy
Project 1: Biophysical human skin cancer model for early skin cancer screening
Watch this 3-min video to understand what is a biophysical model:
Skin cancer is the most common types of malignancy, accounting for over 5.4 million cases and 10 thousand deaths per year in the U.S. Currently, the gold standard for diagnosis of melanoma is visual observation of a suspicious lesion by a dermatologist, followed by a skin biopsy. However, biopsy is painful, inconvenient, and has low diagnostic accuracy.
In this work, I use Raman spectroscopy as an “optical biopsy” method to detect skin cancer fast, noninvasively, and accurately. Additionally, I built a biophysical Raman skin cancer model to better understand the biophysical origin of the skin and skin cancer.
Featured publications:
In this work, I use Raman spectroscopy as an “optical biopsy” method to detect skin cancer fast, noninvasively, and accurately. Additionally, I built a biophysical Raman skin cancer model to better understand the biophysical origin of the skin and skin cancer.
Featured publications:
- Feng X, Moy AJ, Nguyen H, Zhang J, Fox MC, Sebastian KR, Reichenberg JS, Markey MK, Tunnell JW. “Raman active components of skin cancer”, Biomedical Optics Express, 8(6), 2017. DOI: 10.1364/BOE.8.002835
- Feng X, Moy AJ, Nguyen H, Zhang Y, Zhang J, Fox MC, Sebastian KR, Reichenberg JS, Markey MK, Tunnell JW. “Raman biophysical markers in skin cancer diagnosis”, Journal of Biomedical Optics, 23(5), 2018. DOI: 10.1117/1.JBO.23.5.057002
- Zhang Y, Moy AJ, Feng X, Markey MK, Tunnell JW. “Physiological model using diffuse reflectance spectroscopy for non-melanoma skin cancer diagnosis”, Journal of Biophotonics, 2019. DOI: 10.1002/jbio.201900154
- Zhang Y, Moy AJ, Feng X, Nguyen H, Sebastian KR, Reichenberg JS, Markey MK, Tunnell JW. “Assessment of Raman Spectroscopy for Reducing Unnecessary Biopsies for Melanoma Screening”, Molecules, 2020. DOI: 10.3390/molecules25122852
Project 2: Raman spectroscopy for skin cancer surgical margin assessment
Watch this 15-min presentation talk to know about our technology:
Achieving complete tumor resection presents challenges in various cases. While Mohs surgery stands out as the gold standard for nonmelanoma skin cancers, its widespread adoption is hindered by its high cost and infrastructure requirements. The objective of this project is to create a low-infrastructure alternative with Raman spectroscopic imaging, and benefit Mohs-indicated patients in underserved populations.
Featured publications:
Featured publications:
- Feng X, Fox MC, Reichenberg JS, Lopes F, Sebastian KR, Markey MK, Tunnell JW. “Super-pixel averaging Raman spectroscopy for rapid skin cancer margin assessment”, Journal of Biophotonics, 2020. DOI: 10.1002/jbio.201960109
- Feng X, Fox MC, Reichenberg JS, Lopes F, Sebastian KR, Markey MK, Tunnell JW. “A superpixel imaging approach for rapid skin margin assessment.” Molecular-Guided Surgery: Molecules, Devices, and Applications VI. International Society for Optics and Photonics, 2020. DOI: 10.1117/12.2546191
- Feng X, Fox MC, Reichenberg JS, Lopes F, Sebastian KR, Markey MK, Tunnell JW. “Biophysical basis of skin cancer margin assessment using Raman spectroscopy”, Biomedical Optics Express, 10(1), 2019. DOI: 10.1364/BOE.10.000104
- Zhang Y, Moy AJ, Feng X, Nguyen H, Sebastian KR, Reichenberg JS, Markey MK, Tunnell JW. “Diffuse reflectance spectroscopy as a potential method for nonmelanoma skin cancer margin assessment”, Translational Biophotonics, 2020. DOI: 10.1002/tbio.202000001
- Chen M, Feng X, Fox MC, Reichenberg JS, Lopes FC, Sebastian KR, Markey, MK and Tunnell JW, Deep learning on reflectance confocal microscopy improves Raman spectral diagnosis of basal cell carcinoma, Journal of Biomedical Optics, 27(6), p.065004 (2022). DOI: 10.1117/1.JBO.27.6.065004
(2011.8 - 2013.6)
|
Featured publications:
- Feng X, Sun LQ, Zhang EY, ''Depth selectivity for the assessment of microstructure by polarization studies", Biomedical Optics Express, 4(6): 2013. DOI: 10.1364/BOE.4.000958
- Feng X, Sun LQ, Zhang EY, ''Depth selectivity by using polarization-controlled spectral technique", Spectroscopy and Spectral Analysis, 33(11): 2013 (in Chinese). DOI: 10.3964/j.issn.1000-0593(2013)11-2891-04
(2010.10 - 2011.6)
|
(2009.10 - 2010.10)
|
(2009.10 - 2010.7)
|